Biomag - Encyclopedia of Health Conditions - Endoprostheses
Endoprostheses: Definition, Hip, Femur and Other Types

Author MUDr. Peter Bednarčík CSc.
Revision

Do you have long-term and unbearable hip or knee pain? Is it so uncomfortable that it doesn’t let you sleep and conventional medicine doesnt’t work? This is a typical reason for an endoprosthesis. Endoprosthesis means that the damaged or non-functioning joint is replaced with a new one and patients literally return to life.
Find out what the surgery entails, and how best to suppress the symptoms that accompany this procedure. Are there ways to recover as quickly as possible? Read more here.
Indication for an endoprosthesis
- Intense pain
- Limited mobility
- Deteriorating condition
- Inability to carry out daily activities
- Extensive joint damage according to the results of radiological examinations
Each case is individual and the decision to perform an endoprosthesis is made after careful assessment and discussion between the patient and the doctor.

Only a doctor can make a correct diagnosis. Do not use this or any other article on the internet to make a diagnosis. Do not delay seeing a doctor and address your condition early.
Description and causes of the need for implantation of an endoprosthesis
What is an endoprosthesis?
An endoprosthesis is an artificial joint that is inserted during surgery to replace a damaged joint. The most common joints operated on are the hip joint, knee joint and shoulder. The term total joint replacement (TEP) means that the entire joint is replaced.
Artificial joint replacement is usually the solution for patients who have a severe form of arthritis or other joint disease. Conservative non-surgical treatment had no effect and the pain did not subside even after pain medication. An endoprosthesis usually means a definitive solution to the problem with the diseased joint, as a total hip replacement or total knee replacement can last 20 years, and often longer.
Endoprosthesis is generally recommended when the cartilage in the joint is so damaged that the joint cannot function. [1]
Types of endoprostheses
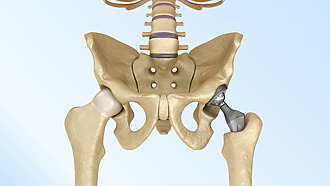
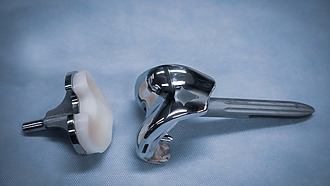
Hip endoprosthesis
It is a surgical solution for advanced arthrosis of the hip. It is also used for severe rheumatoid arthritis (inflammatory joint disease), after some fractures or for osteonecrosis (bone death). Surgery is usually reserved for patients who:
- suffering from long-term hip pain,
- have limited hip mobility and difficulty walking,
- report sleep disturbances due to pain. [2]
Total hip replacement means that both the femoral head and the hip socket are replaced. [3]
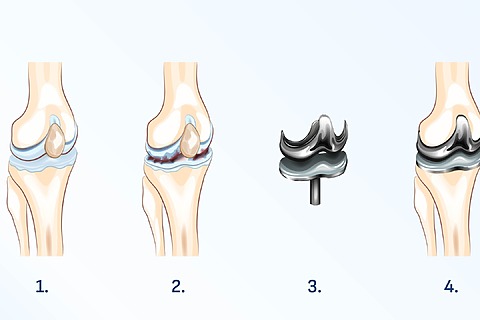
Endoprosthesis of the knee
This is an operation to replace a damaged knee joint with an artificial one. The most common reason for surgery is severe knee arthritis, rheumatoid arthritis or a previous injury to the joint.
- The goal of the surgery is to relieve pain, swelling and restore mobility in the knee.
- Total knee arthroplasty will enable patients to walk again without difficulty and to engage in normal daily activities. [4]
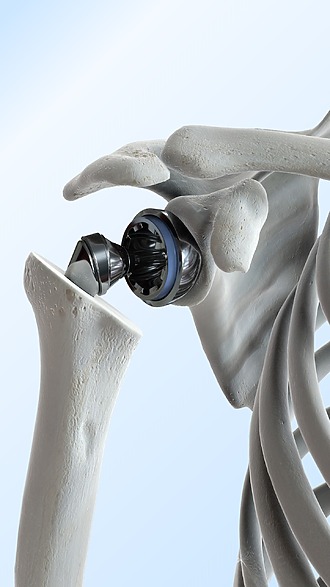
Shoulder joint endoprosthesis
It represents the surgical treatment of advanced arthrosis of the shoulder. Depending on the degree of damage, partial or total joint replacement is chosen.
- In addition to arthrosis, indications for total arthroplasty include rheumatoid arthritis or osteonecrosis.
- The shoulder joint can also be damaged due to a muscle injury in the shoulder area or a fracture of the humerus.
- Thanks to the operation, many patients get rid of shoulder pain completely. Most of them report better range of motion and greater strength. [5]
Preparing for surgery
Hip surgery – total hip replacement and other surgical procedures with artificial joints require preparation. Pre-operative examination includes, among other things, blood tests and X-rays. At the hospital, patients usually have a consultation with the surgeon who will operate on them. Immediately before surgery, it is necessary to follow the prescribed regimen (eating and drinking on the day of surgery, possibly discontinuing certain medications). It is advisable to be in good condition for the operation to ensure that the procedure and recovery are as smooth as possible. [6]
Total endoprosthesis - surgery
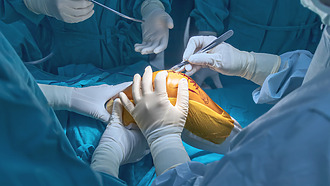
Total hip arthroplasty and other surgeries where an artificial joint is inserted into the body usually take several hours. During the operation, the surgeon removes the damaged parts of the joint (cartilage, bone) and replaces them with artificial materials. Metals, plastics and ceramics are used. [7]
- Hip surgery is performed under either partial or general anaesthesia. The surgeon makes an incision in the hip area to access the entire joint.
- The knee and shoulder joint is usually operated on under general anaesthesia. This is an open operation in which the knee or shoulder area is cut open.
- After surgery, the patient is monitored in the intensive care unit followed by transfer to a regular hospital ward.
- The length of hospital stay depends on the type of surgery and the patient’s general condition. It usually takes days. [8]
Did you know?
- In the Czech Republic, more than 200 hip surgeries per 100,000 inhabitants are performed every year. Germany leads the European statistics with more than 300 operations.
- The number of artificial joint replacements operated on has increased significantly since 2009. This is due to the increased incidence of arthritis and the ageing population. [9]
Complications associated with endoprosthesis
Both knee and hip arthroplasty can have various complications. Surgery is associated with the risk of:
- blood clots, surgical site infections or nerve injuries,
- loosening or displacement of the endoprosthesis into the wrong position,
- different limb lengths, which can result from muscle tightness in the hip area,
- reoperation.
Blood clot
Infection
Loosening of endoprothesis
Reoperation

We recommend not delaying treatment
Do not delay treatment , and see a specialist if you have any health problems or doubts about your health. This will prevent unnecessary health complications.
Treatment - What happens after surgery
Shortly after surgery, you need to start moving the new joint. Movement serves as a prevention of blood clots that can occur after surgery. The first movements and getting out of bed are supervised by a physiotherapist who will also recommend further exercises. Exercise is important during the hospital stay and should be continued long term after discharge home.
- Patients receive specific instructions from the doctor on how to gradually load the operated joint and what movements to avoid.
- Proper positioning of the limb (elevated position) and cool compresses help reduce swelling that may occur after surgery. [12]
Pain advice: patients should only take pain medication that has been recommended by their doctor. Some over-the-counter pain medications may increase the risk of bleeding. [13]

Beware
Treatment after an endoprosthesis can only be determined by your doctor after considering your overall health. Therefore, do not use this article as a guide to treatment, which can only be determined by your doctor.
Recovery at home
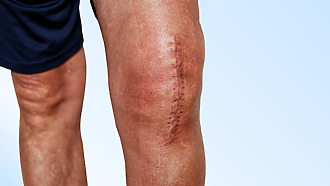
After arriving home from the hospital, you need to adapt the environment to the current possibilities of movement. How to minimize the risk of a fall that could damage the operated joint?
- Use crutches or a cane as directed by your doctor and physiotherapist.
- Remove loose carpets and cables that you may trip over.
- Keep all the necessary items close at hand so you don’t have to bend down or reach up.
- Use non-slip aids when showering.
- Use the raised toilet seat attachment.
- Try dressing aids such as a sock threader.
After surgery, it is necessary to take care of the scar. The patient should learn the correct technique of scar massage in the hospital. If swelling, bleeding, redness, or worsening pain occurs at the surgical wound after arriving home, a doctor should be consulted. [14]
Long-term results with endoprosthesis
Most patients make a full recovery after total knee arthroplasty. They get rid of pain, the operated joint regains mobility and the quality of life improves significantly.[15] The speed and rate of recovery is influenced by:
- overall physical condition before surgery,
- the severity of the joint disease,
- the scope and type of operation,
- quality of rehabilitation. [16]
Total hip arthroplasty durability: patients can expect the new joint to last 20 years or more.
Professional statistics indicate that the need for revision for total hip and knee arthroplasty is around 6% at five years after surgery and 12% at ten years after surgery. [17]
Alternative treatment methods
Total joint arthroplasty may not be an appropriate treatment for arthritis or other joint disease in some people. Physiotherapy, anti-inflammatory drugs, corticosteroids or dietary supplements known as joint nutrition may provide relief from symptoms such as pain or joint stiffness. Surgery, in which the damaged joint is only modified, not completely replaced, can also be a solution. [18]
Lifestyle after arthroplasty
Most patients return to their lifestyle three months after surgery. Gradual improvement continues throughout the first year after surgery. [19]
- Returning to work is usually possible after a few weeks, depending on the type of work and the extent of the surgery.
- Wait a few months to drive a car until the operated limb is strong and stable enough.
- Swimming is recommended after hip surgery; on the contrary, tennis or running puts excessive strain on the new joint. [20]
Total hip arthroplasty - spa treatment

Spa treatment after total hip, knee or shoulder arthroplasty is of great importance. The combination of physiotherapy and therapeutic baths promotes the mobility of the new joint and strengthens the weakened muscles. Many patients need to adjust their whole body posture as they have been seeking different positions of relief during prolonged joint pain.
- Spa care should be started within 3 months of surgery.
- Patients can also get to the spa directly from the hospital. This is called bed-to-bed transfer.
- The spa provides comprehensive rehabilitation including gait training.
- During the spa stay it is also advisable to take care of the scar – for example, phototherapy is used. [21]

Important information with endoprosthesis
Total joint replacement is a surgical solution for advanced arthrosis or other disease that significantly impairs the functionality of the joint.
The damaged joint is replaced with an artificial joint replacement.
After surgery, careful and sufficient rehabilitation is required.
Summary and recommendations for managing pain
See your Physician
Treatment after joint replacement is always determined by your doctor based on a general examination, an assessment of your condition and an accurate diagnosis.
Causal Treatment of Pain
After a general examination, your doctor will recommend treatment. In connection with this, they will also recommend possible lifestyle modification and further courses of action.
Relieving pain, promoting healing and blood circulation
Symptomatic treatment focuses on the manifestations or symptoms of the disease. Such treatment can significantly improve your quality of life and support comprehensive treatment as the disease progresses.
Sources, references and literature
[1] Shah B. Joint Replacement Surgery. American College of Rheumatology. February 2023. Available at: https://rheumatology.org/patients/joint-replacement-surgery
[2] Hip Replacement. Mayo Clinic. 22. April 2022. Available at: https://www.mayoclinic.org/tests-procedures/hip-replacement/about/pac-20385042
[3] Total Joint Replacement. American Academy of Orthopaedic Surgeons. Available at: https://orthoinfo.aaos.org/en/treatment/total-hip-replacement/
[4] Knee Replacement Surgery Procedure. Johns Hopkins Medicine. Available at: https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/knee-replacement-surgery-procedure
[5] Shoulder replacement surgery. Mayo Clinic. 10. May 2023. Available at: https://www.mayoclinic.org/tests-procedures/shoulder-replacement/about/pac-20519121
[6] Hip Replacement. Mayo Clinic. 22. April 2022. Available at: https://www.mayoclinic.org/tests-procedures/hip-replacement/about/pac-20385042. Total Joint Replacement. American Academy of Orthopaedic Surgeons. Available at: https://orthoinfo.aaos.org/en/treatment/total-hip-replacement/
[7] Total Joint Replacement. American Academy of Orthopaedic Surgeons. Available at: https://orthoinfo.aaos.org/en/treatment/total-hip-replacement/
[8] Hip Replacement. Mayo Clinic. 22. April 2022. Available at: https://www.mayoclinic.org/tests-procedures/hip-replacement/about/pac-20385042. Shoulder replacement surgery. Mayo Clinic. 10. May 2023. Available at: https://www.mayoclinic.org/tests-procedures/shoulder-replacement/about/pac-20519121
[9] Hip and Knee Replacement. Health at a Glance 2021: OECD Indicators. OECD iLibrary.Available at: https://www.oecd-ilibrary.org/sites/8b492d7a-en/index.html?itemId=/content/component/8b492d7a-en
[10] Total Joint Replacement. American Academy of Orthopaedic Surgeons. Available at: https://orthoinfo.aaos.org/en/treatment/total-hip-replacement/
[11] Knee Replacement Surgery Procedure. Johns Hopkins Medicine. Available at: https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/knee-replacement-surgery-procedure
[12] Knee Replacement Surgery Procedure. Johns Hopkins Medicine. Available at: https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/knee-replacement-surgery-procedure. Hip Replacement. Mayo Clinic. 22. April 2022. Available at: https://www.mayoclinic.org/tests-procedures/hip-replacement/about/pac-20385042
[13] Knee Replacement Surgery Procedure. Johns Hopkins Medicine. Available at: https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/knee-replacement-surgery-procedure
[14] Knee Replacement Surgery Procedure. Johns Hopkins Medicine. Available at: https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/knee-replacement-surgery-procedure
[15] Total Joint Replacement. American Academy of Orthopaedic Surgeons. Available at: https://orthoinfo.aaos.org/en/treatment/total-hip-replacement/
[16] Shah B. Joint Replacement Surgery. American College of Rheumatology. February 2023. Available at: https://rheumatology.org/patients/joint-replacement-surgery
[17] Labek G., Thaler M., Janda W., Agreiter M., Stöckl B. Revision rates after total joint replacement: cumulative results from worldwide joint registry datasets. J Bone Joint Surg Br. 2011 Mar; 93 (3): 293-297, doi: 10.1302/0301-620X.93B3.25467. Erratum in: J Bone Joint Surg Br. 2011 Jul; 93 (7): 998. Available at: https://pubmed.ncbi.nlm.nih.gov/21357948/
[18] Cluett J. Alternatives to Hip Replacement. Verywell Healths. 7. March 2022. Available at: https://www.verywellhealth.com/alternatives-to-hip-replacement-2549516
[19] Hip Replacement. Mayo Clinic. 22. April 2022. Available at: https://www.mayoclinic.org/tests-procedures/hip-replacement/about/pac-20385042
[20] Activities After Total Hip Replacement. American Academy of Orthopaedic Surgeons. Available at: https://orthoinfo.aaos.org/en/recovery/activities-after-hip-replacement/
[21] Conditions after orthopaedic surgeries and joint replacements. Spa Belohrad. Available at: https://belohrad.cz/nejcasteji-lecime/stavy-po-ortopedickych-operacich-a-kloubnich-nahradach. TEP – spa after joint replacement. Darkov Spa. Available at: https://www.laznedarkov.cz/lazne-po-vymene-kloubu
| Rate this article |
|
|
5/52 Reviews
|
How to control pain, promote healing and blood circulation?
The solution may be symptomatic treatment with pulsed magnetic therapy, which targets symptoms and signs such as pain, inflammation and impaired blood flow.In addition, such treatment can support comprehensive treatment and significantly improve quality of life.
Explaining the effects
The basic principle of Biomag 3D pulsed magnetic therapy (PEMF therapy) is the generation of electromagnetic pulses. These pulses penetrate through clothing and through the entire depth of the tissue to the point of targeted application. The pulses have specially developed biotropic parameters (e.g. frequency, shape, intensity) to best affect various health problems
What are the effects of 3D magnetic therapy (PEMF therapy)?
- Helps relieve pain.
- Mitigates inflammation.
- Promotes blood circulation.
How is the treatment applied?
The application is very simple. You select the desired therapeutic effect on the device and attach the supplied applicator to the desired application site. Magnetic therapy is usually applied 2 times a day for 20 minutes.
We will be happy to help you try this method and advise you on which device to purchase.